This review was conducted as part of an update of the NICE COPD guideline (NG115). The search strategies used in this review are detailed in appendix C.ĭeclarations of interest were recorded according to NICE’s 2018 conflicts of interest policy. To try to assess the effect of including 2 different overlapping cut offs in each subgroup, a sensitivity analysis was carried out removing the study using 200 cells per microlitre as a cut-off ( Singh 2016 for triple therapy versus LABA+ICS, Papi 2018 for triple therapy versus LAMA+LABA). 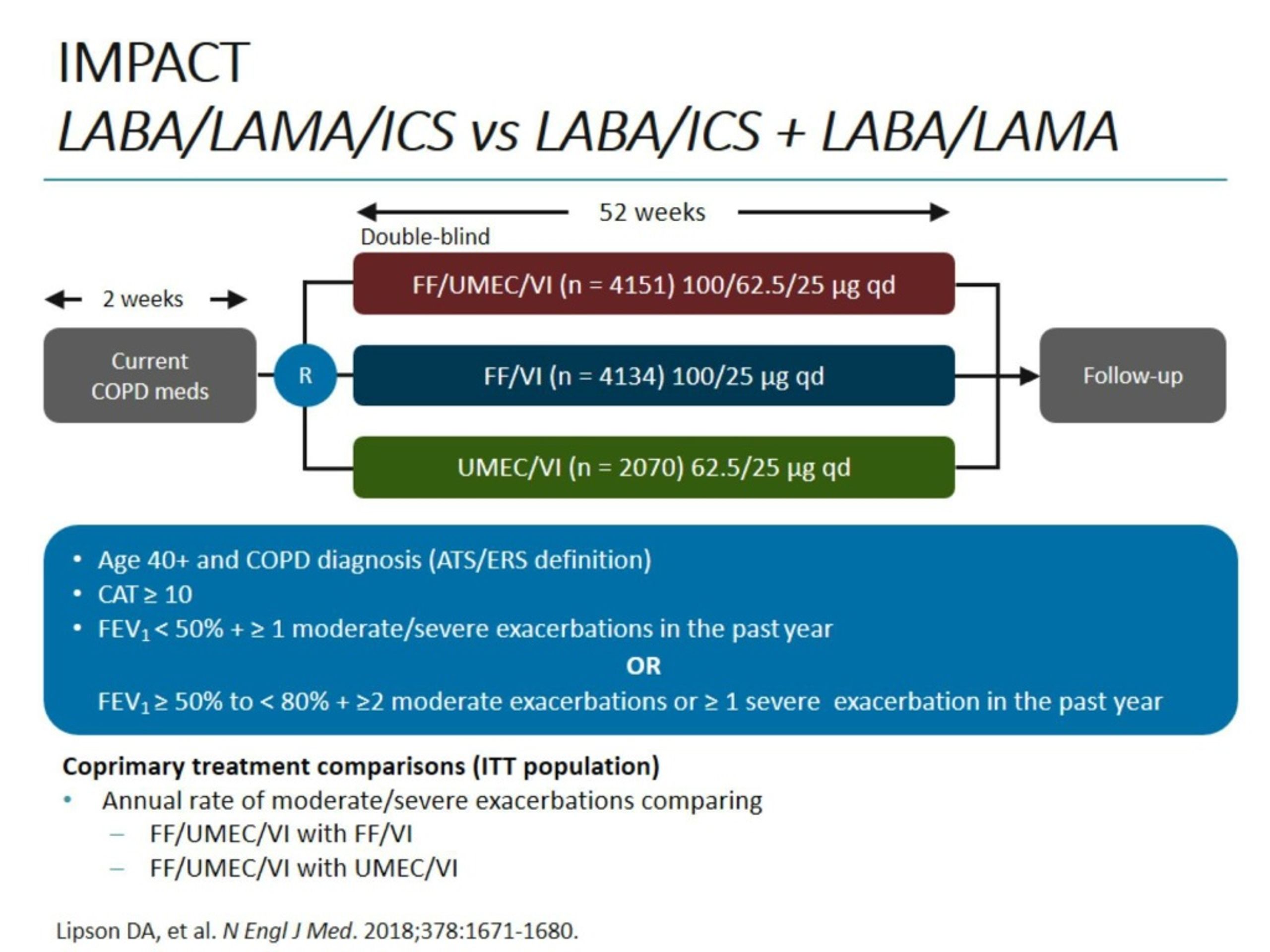
As a result, eosinophil count subgroups were separated into ‘higher eosinophil count per microlitre including trials with cut offs of greater than 150 or 200 eosinophils per microlitre’ and ‘lower eosinophil counts per microlitre’ for studies reporting less than 150 or 200 eosinophils per microlitre. Different studies separated people by different eosinophil count thresholds, some by those above or below 200 cells per microliter and others by those above or below 150 cells per microliter. However, sub-group analyses for inhaler type, exacerbation history, prior medication and eosinophil count were carried out. It was not possible to separate whole studies or groups of participants within studies by variation in baseline peak flow, FEV1 variability, asthma, smoking status or pulmonary rehabilitation completion status. No data was available to perform some of the pre-specified sub-group analyses. The protocol for the review is summarised in Table 1. Studies which specifically compared the effectiveness of triple therapy alone using a single inhaled device or using separate inhalers were not eligible for inclusion in this review. Single and multiple inhaler doses of triple therapy were included as separate subgroups in the analyses in this review, but the main comparison of interest was between the effects of dual and triple therapy, rather than inhaler type. This review aimed to evaluate the effectiveness of triple therapy, either delivered as a combination of inhalers, or as one single inhaler, in managing the symptoms of patients with severe COPD in comparison to the dual therapy combinations of LAMA+LABA and LABA+ICS. Triple therapy can be prescribed as a single inhaler which delivers all three drugs in one dose or as multiple inhalers which deliver separate doses of each drug. ‘Triple therapy’ is delivery of a combination of all three inhaled drugs (LAMA+LABA+ICS). Possible combinations include long-acting muscarinic antagonist with long-acting beta-adrenoceptor (LAMA+LABA) or LABA with inhaled corticosteroids (LABA+ICS). 
Inhaled drugs are often used in combination to provide more effective relief. The treatment of moderate to very severe COPD commonly includes the use of long-acting bronchodilators and inhaled corticosteroids to ease symptoms and reduce exacerbations.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
